Cupric Oxide
In stock
SKU
C6023
Cupric Oxide
| Description | 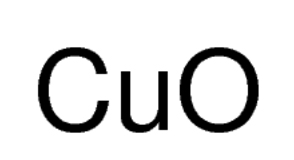 Uses: Cupric oxide has a variety of uses including optical glass polishing agent, wood preservative, pigment, agricultural chemical and abrasive, and welding flux for bronze materials. It is used in the production of intermediates, oxidizing/reducing agents, process regulators and surface treating agents. It finds use in the manufacture of rayon, phosphors and dry battery cells. It’s used as a catalyst in certain chemical reactions of industrial importance. It is also used in chemical sensors and superconducting materials. Through oxidation with cupric oxide, hazardous materials like cyanides, dioxins and halogenated hydrocarbons can be disposed. |
|---|---|
| Chemical Synonym | Copper (II) oxide |
| SKU | C6023 |
| CAS No. | 1317-38-0 |
| Property 1 | 99%, black |
| Property 2 | purified |
| Handling and Storage | Precautions for safe handling
Avoid contact with skin and eyes. Avoid formation of dust and aerosols. Further processing of solid materials may result in the formation of combustible dusts. The potential for combustible dust formation should be taken into consideration before additional processing occurs. Provide appropriate exhaust ventilation at places where dust is formed. Conditions for safe storage, including any incompatibilities: Keep container tightly closed in a dry and well-ventilated place. Recommended storage temperature 2 - 8 °C Keep in a dry place. |
| Transport Information | DOT (US) Not dangerous goods IMDG UN number: 3077 Class: 9 Packing group: III EMS-No: F-A, S-F Proper shipping name: ENVIRONMENTALLY HAZARDOUS SUBSTANCE, SOLID, N.O.S. (Cupric oxide) Marine pollutant: yes IATA UN number: 3077 Class: 9 Packing group: III Proper shipping name: Environmentally hazardous substance, solid, n.o.s. (Cupric oxide) |

